Enamel remineralization is the natural process of restoring essential minerals like calcium and phosphate back into your tooth’s protective crystal matrix. By using biomimetic ingredients like Nano-Hydroxyapatite or Fluoride and maintaining a neutral oral pH (above 5.5), you can effectively harden weakened enamel, reverse early-stage “white spot” decay, and significantly reduce tooth sensitivity.
Introduction to Enamel Remineralization
When we think of our teeth, we often imagine them as static, bone-like structures—permanent fixtures that simply “are.” However, from a clinical perspective, your enamel is one of the most dynamic tissues in the human body. It is constantly engaged in a microscopic tug-of-war.
Every time you take a sip of coffee, enjoy a citrus salad, or snack on crackers, a chemical reaction begins. This guide is designed to take you beyond the surface of “brushing and flossing” and into the sophisticated science of enamel remineralization. By the end of this article, you will understand how to shift your oral chemistry from a state of decay to a state of self-repair.
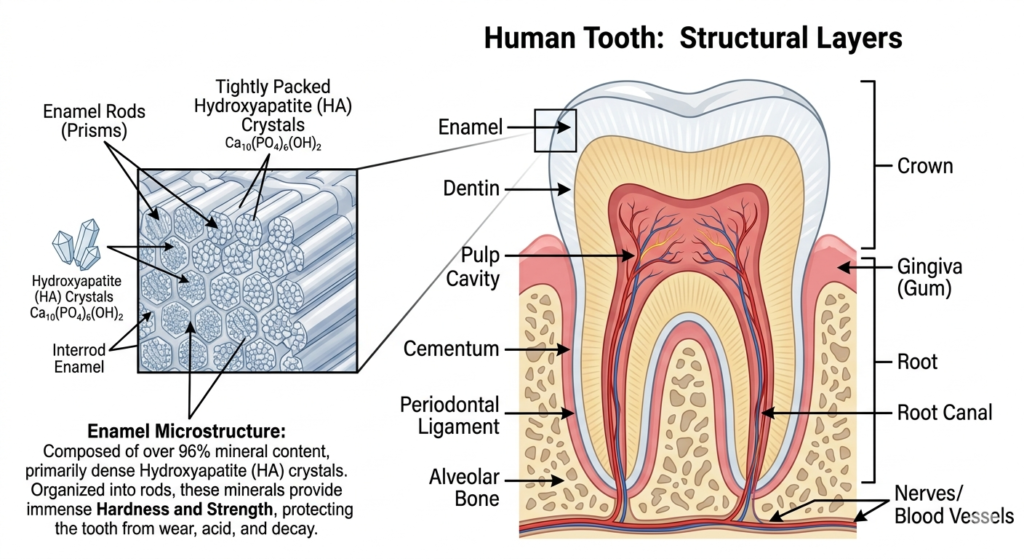
What Exactly is Enamel?
To understand how to fix enamel, we must first understand its composition. Enamel is the most highly mineralized substance in your body, even harder than bone. It is composed of roughly 96% mineral, primarily a crystalline form of calcium phosphate called hydroxyapatite.
The Anatomy of the Crystal Matrix
Unlike your skin or bones, enamel contains no living cells. This means it cannot “grow back” in the traditional sense once it is physically chipped or worn away. However—and this is the key to modern preventative dentistry—the mineral matrix can be reinforced and rebuilt if the structural “scaffolding” is still present. This is the essence of remineralization.
Why Enamel Thins Over Time
Enamel erosion is often a “silent” process. It happens through mechanical wear (attrition), chemical dissolution (erosion), or bacterial attack (caries). As the enamel thins, the yellowish dentin underneath begins to show through, leading to a duller smile and increased sensitivity.
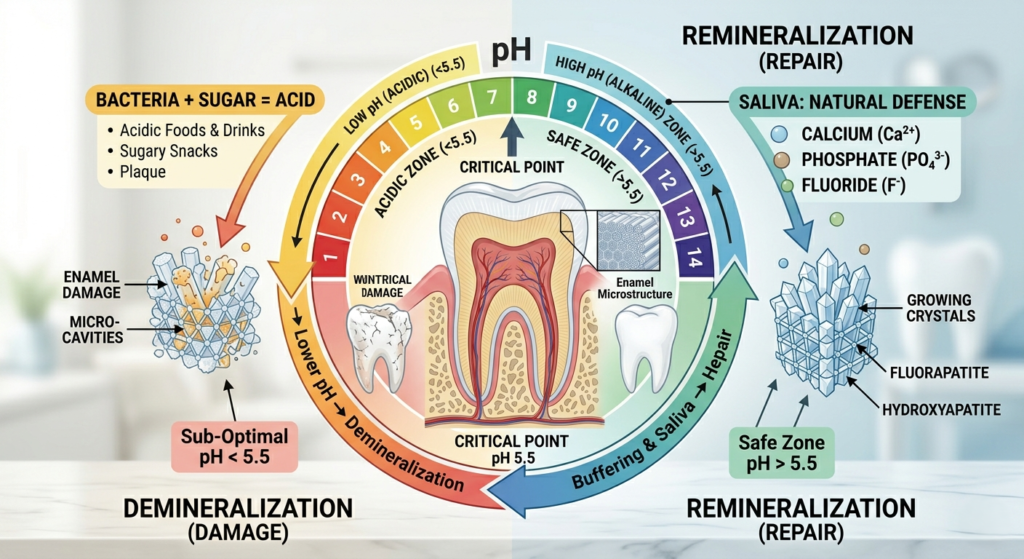
The Tug-of-War: Demineralization vs. Enamel Remineralization
Your mouth is a chemistry lab. The process of tooth decay is not an event; it is a continuous cycle that resets every time you eat or drink.
The Demineralization Phase
When you consume fermentable carbohydrates (sugars and starches), the bacteria in your oral microbiome—specifically Streptococcus mutans—metabolize those sugars and excrete acid as a byproduct.
- The Critical pH: Once the pH level in your mouth drops below 5.5, the environment becomes acidic enough to dissolve the hydroxyapatite crystals.
- Mineral Leaching: Calcium and phosphate ions are pulled out of the enamel and into the saliva. If this continues, the enamel becomes porous, leading to “white spot lesions.”
The Enamel Remineralization Phase
Once the acid is neutralized, the reverse happens. Your saliva, which is naturally rich in calcium and phosphate, acts as a delivery vehicle.
- The Role of Saliva: Saliva is your body’s natural defense mechanism. It contains bicarbonate buffers that neutralize acid and statherin proteins that keep calcium in a state ready for your teeth to absorb.
The Heavy Hitters: Fluoride vs. Nano-Hydroxyapatite
In the world of SEO and dental health, the “Fluoride vs. nHAp” debate is a trending topic. Let’s look at the evidence-based reality of both through a clinical lens.
Fluoride: The Industrial Guard
Fluoride works through a process called ion substitution. When fluoride is present during remineralization, it replaces the hydroxyl group in the hydroxyapatite crystal. This creates Fluorapatite.
- The Advantage: Fluorapatite has a critical pH of 4.5, making it more resistant to acid than your original enamel.
- The Drawback: Fluoride primarily works on the surface level. It creates a hard “shell” but may not always address deep-tissue porosity.
Nano-Hydroxyapatite (nHAp): The Biomimetic Future
Originally developed by NASA to help astronauts maintain bone and tooth density in zero gravity, nHAp is the “gold standard” for the “Calm Dentist” philosophy.
- How it Works: Because the particles are “nano-sized,” they can fit into the microscopic tubules and pores of the enamel. It doesn’t just create a shield; it acts as a filler, physically replacing the lost mineral with an identical substance.
- Safety: nHAp is biocompatible and non-toxic, making it an excellent choice for patients looking for “fluoride-free” alternatives that actually work.
Dietary Biohacking for Stronger Enamel
You cannot out-brush a bad diet. To support remineralization, your body needs the systemic building blocks to produce high-quality, mineral-dense saliva.
The Vitamin D3/K2 Connection
Vitamin D acts as the “gatekeeper” for calcium absorption. However, without Vitamin K2, that calcium may end up in your arteries instead of your teeth. K2 activates osteocalcin, which pulls calcium into the dental matrix.
Phytic Acid and Mineral Absorption
Found in grains and legumes, phytic acid can “bind” to minerals, making them unavailable for your body to use. If you have thin enamel, consider soaking or sprouting your grains to reduce phytic acid and improve mineral bioavailability.
The Importance of Arginine
This amino acid, found in many proteins, helps foster a “base-producing” microbiome. This keeps your oral pH higher for longer periods, extending the window of time where remineralization can occur.
Enamel Repair
If you are noticing transparency at the edges of your teeth or sharp pain when drinking cold water, your remineralization cycle is lagging. Here is a clinical protocol to reset it.
Step 1: Alkalize Immediately
After eating, rinse with water or a baking soda solution (1/4 tsp in water) to immediately jump from an acidic pH back to a safe 7.0. This stops demineralization in its tracks.
Step 2: Choose Your Vehicle
Use a toothpaste containing at least 10% nano-hydroxyapatite. If you prefer fluoride, ensure it is a high-quality formulation that isn’t excessively abrasive (low RDA value).
Step 3: Mouth-Breathing Correction
If you sleep with your mouth open, your teeth dry out. Without saliva, remineralization stops. Consider using a humidifier or speaking to a professional about airway health and nasal breathing.
Step 4: The “Spit, Don’t Rinse” Rule
After brushing with a remineralizing paste, do not rinse with water. Let the residue sit on your teeth. This allows the calcium and phosphate ions time to penetrate the enamel pores.
Professional Interventions at the Clinic
Sometimes, at-home care isn’t enough to reverse deep structural loss. In our practice, we utilize advanced biomimetic techniques.
Silver Diamine Fluoride (SDF)
SDF is a liquid that “freezes” decay instantly. It is particularly useful for sensitive areas or early-stage cavities that we want to manage without drilling. The only drawback of this topical agent is that it stains the decayed tooth structure black.
Resin Infiltration (Icon)
This is a revolutionary way to fill white spot lesions. We use a high-penetration resin to fill the pores of the enamel, restoring the tooth’s natural color and strength without any anesthesia.
Professional Topical Pastes
We often prescribe high-concentration calcium-phosphate delivery systems (like MI Paste) for patients with chronic dry mouth or high decay rates.
FAQs (People Also Ask)
Can enamel grow back?
While you cannot grow “new” enamel once the tooth has fully formed, you can remineralize existing enamel. This process makes the tooth harder, thicker, and significantly more resistant to future acid attacks.
Enamel Remineralization: How long does it take to remineralize teeth?
Microscopic repair begins immediately, but visible improvements—such as a reduction in sensitivity or the fading of white spots—typically take 3 to 4 months of consistent mineral application and pH management.
Is charcoal toothpaste good for enamel?
No. Charcoal is highly abrasive. While it may remove surface stains, it can physically scrub away the thin layer of enamel you are trying to save. We recommend sticking to biomimetic ingredients like nHAp.

Dr. Mehmood Asghar is a Dentist and an Assistant Professor of Dental Materials, with nearly a decade of clinical experience. He holds a Ph.D. in Dental Biomaterials from Universiti Malaya and a Bachelor of Dental Surgery (BDS) from NUST. As a dedicated researcher with over 25 peer-reviewed publications and more than 310 citations in international journals, Dr. Asghar bridges the gap between clinical dentistry and advanced biomaterials research to provide patients with evidence-based, practical dental advice.

